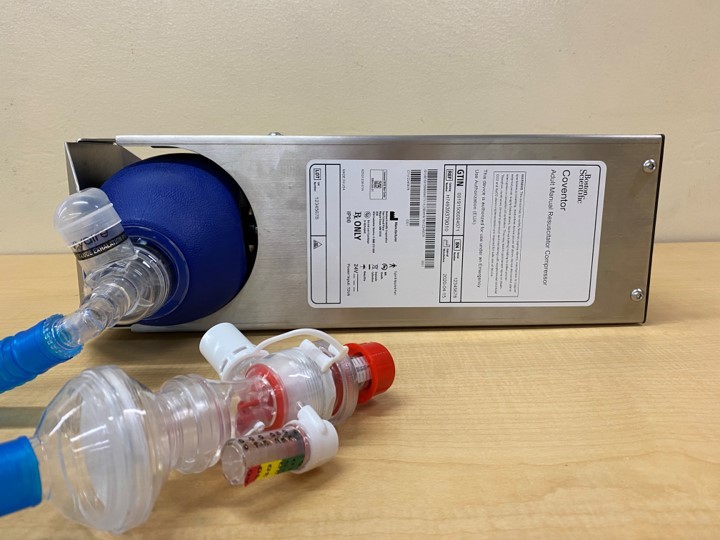
In response to COVID-19, the Earl E. Bakken Medical Devices Center designed and built the first FDA Emergency Use Approved ventilator alternative known as the Coventor. A multidisciplinary team of physicians, engineers, and students created a compact, low-cost, easily manufactured ventilator system that uses readily available components familiar to clinicians. With the pandemic causing significant supply chain issues, the Coventor’s distinct advantage was that it didn’t rely on traditional ventilation system components. Sixty-seven days after the onset of COVID-19 in the US, the UMN team collaborated with local and national industry leaders including Boston Scientific, Medtronic, and UnitedHealth Group to manufacture 3,000 Coventor units for immediate deployment. These units were given to the national stockpile and non-profit organizations worldwide. With commercial manufacturing of the Coventor in place, the team continues to provide support to users, engineers, and clinicians around the globe. Additionally, the Coventor design was released as open-source and has been downloaded for use in over 20 countries. Your gift will benefit the Earl E. Bakken Medical Devices Center educational, research, and outreach programs, with the current priority being continued research and enhancements of ventilator systems.


https://crowdfund.umn.edu/campaign/Coventor-A-COVID-19-Ventilation-System